What is in a atom? What makes up an element?
We have spent the last few days talking about what makes up atoms. We have visualized atoms using our bean models, and we have talked about what makes elements react.
Our first lab on chemical reactivity will be due on Thursday.
In this lab you first take and describe the four metals you had out for the group. Magnesium, Calcium, Tin, and Aluminum. It is important to describe them well because changes in their appearance may be the only way you know a chemical reaction is taking place.
For the first trial you need to cover the metals with water, and have them sit for 4 minutes to see if there is a reaction.
You need to observe their appearance to see if it is the same or different than before adding the water.
For the second trial you will need to get a new sample of all of the metals.
You will then cover each metal with acid, and observe the results. Allow it to sit for 4 minutes, and then write down any changes.
You will submit a full lab write up for this lab.
Second Lab
For this lab we are going to be looking at replacement reactions. That is when a metal reacts with a solution does the metal stay or does it switch places with part of the solution.
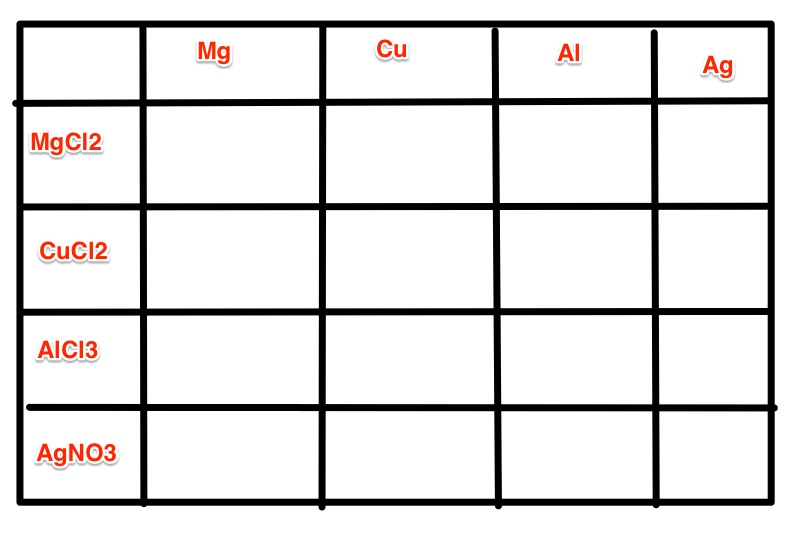
For this lab you will get a multi-well plate. You need to fill the columns going down with the same metal. So if Copper is going to go in the first well it will go in the three wells directly below it. The metals will be Mg, Cu, Al, Ag.
You need to describe all of the metals before you add the liquids.
Next you will need to add the solutions. Now every solution is going to go across the wells. So if CuCl2 is in the second row all of the wells in the second row need to be filled with CuCl2. You need to describe the look of all of the solutions before you add them to the metals.
After they sit please mark any of the wells that either the metal or the solution has changed in, and how it has changed.
Our first lab on chemical reactivity will be due on Thursday.
In this lab you first take and describe the four metals you had out for the group. Magnesium, Calcium, Tin, and Aluminum. It is important to describe them well because changes in their appearance may be the only way you know a chemical reaction is taking place.
For the first trial you need to cover the metals with water, and have them sit for 4 minutes to see if there is a reaction.
You need to observe their appearance to see if it is the same or different than before adding the water.
For the second trial you will need to get a new sample of all of the metals.
You will then cover each metal with acid, and observe the results. Allow it to sit for 4 minutes, and then write down any changes.
You will submit a full lab write up for this lab.
Second Lab
For this lab we are going to be looking at replacement reactions. That is when a metal reacts with a solution does the metal stay or does it switch places with part of the solution.
For this lab you will get a multi-well plate. You need to fill the columns going down with the same metal. So if Copper is going to go in the first well it will go in the three wells directly below it. The metals will be Mg, Cu, Al, Ag.
You need to describe all of the metals before you add the liquids.
Next you will need to add the solutions. Now every solution is going to go across the wells. So if CuCl2 is in the second row all of the wells in the second row need to be filled with CuCl2. You need to describe the look of all of the solutions before you add them to the metals.
After they sit please mark any of the wells that either the metal or the solution has changed in, and how it has changed.