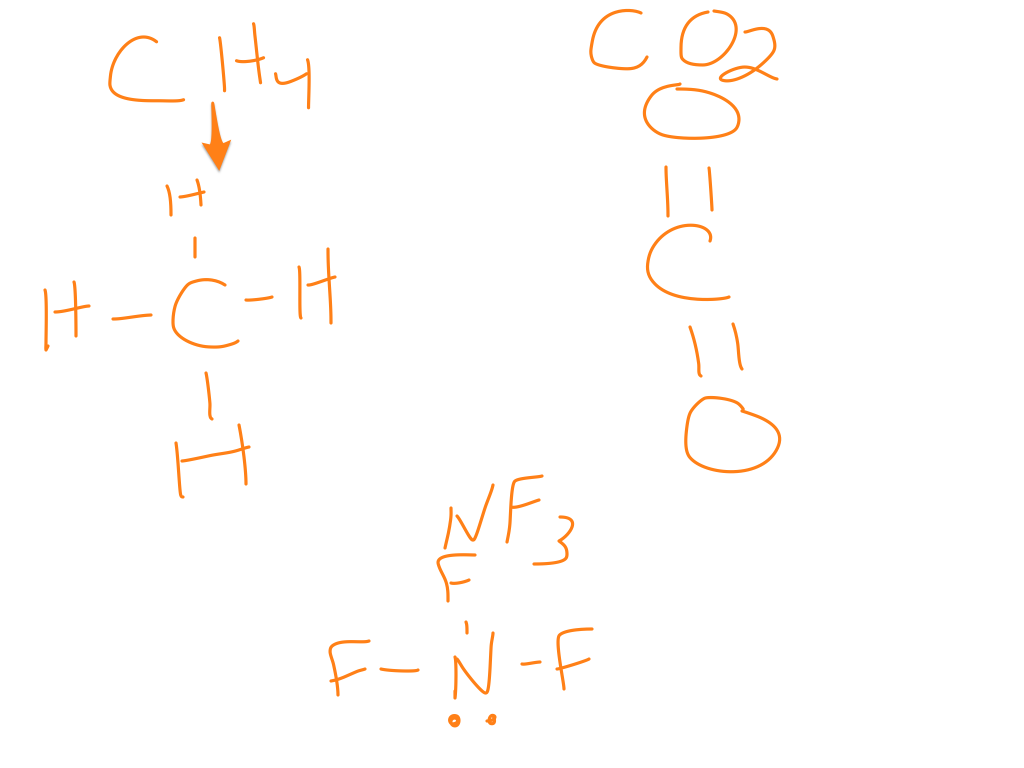Covalent bonds. Making Compounds
Page 244 1-5 due Friday 23rd
Chapter 8 Notes
|
The links are posted for the lab (the .docx)we will do on Monday 26th and the actual shapes for the medicines (the .pptx)
|
|
| ||||||||||||
Click on the link to go to Socrative
My room number is 372602
My room number is 372602
|
|
|
|
The worksheet with molecular structures. Is Due Wed. 29th.
To the right are some examples of normal molecular structures.
| |||||||
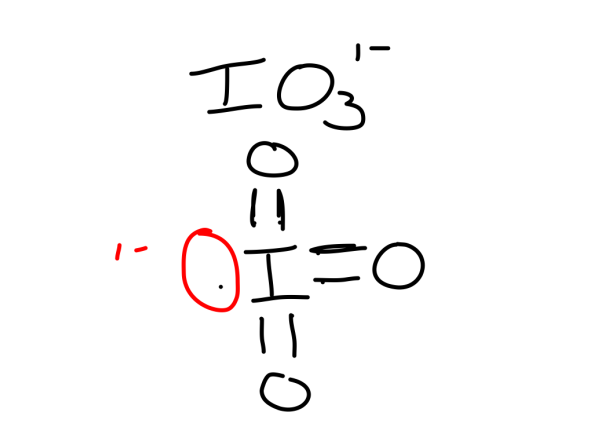
A polyatomic ion which shows odd electron, and expanded octets.
|
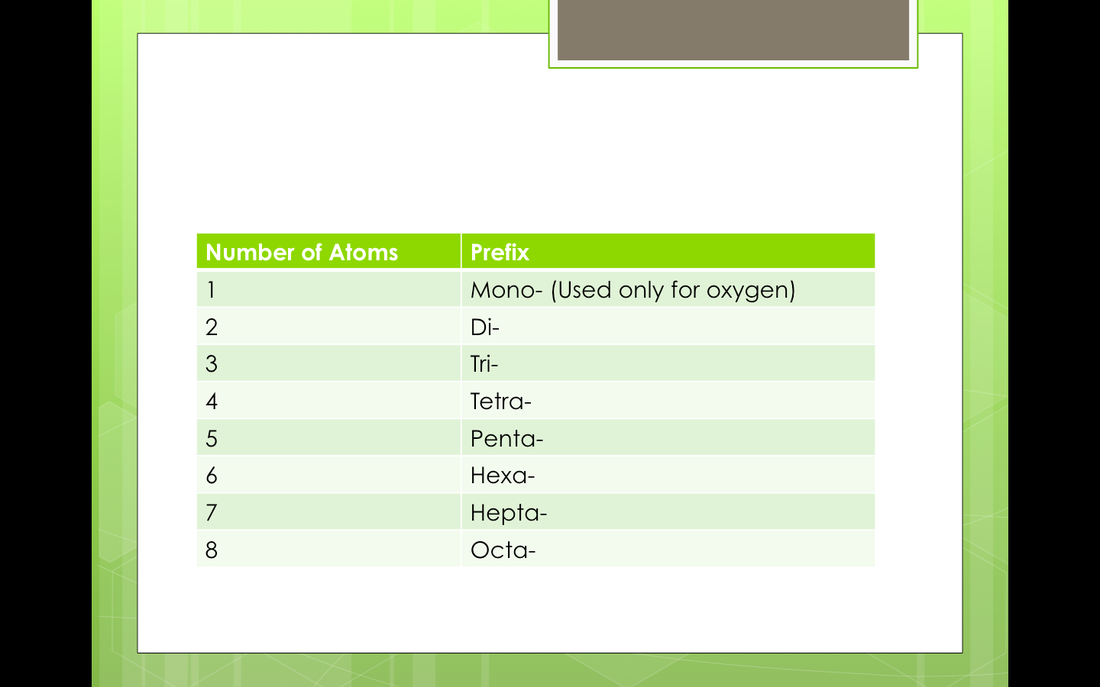
We will be going over naming multiple days in class. There are a few different rules depending on what the covalent bond is making. When we finish you will have to turn in the page to your right. You will summit this one electronically because you will only need to write the names not draw the structures
|
| ||||||
|
This is an extra of the Chemical bonding lab that we will do.
| ||||||
Naming Gases- Any mixture of gases if the vapor point stays below room temperature will result in a gas being formed. It isn't needed to state that mixtures of gases are still in gas form in the way they are named. However, we denote elements in gas form as such:
Name the element and follow it by gas. He is Helium gas, and O2 is Oxygen gas. When you see an elemental gas written all non-noble gases are paired up and Noble gases are stand alone.
Name the element and follow it by gas. He is Helium gas, and O2 is Oxygen gas. When you see an elemental gas written all non-noble gases are paired up and Noble gases are stand alone.