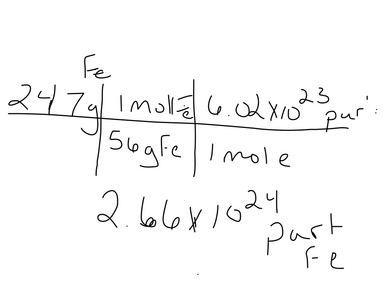The Mole is a unit
We use it to generalize how many atoms are in one group. Because they are so small we have a very large number, but it is the same idea as saying that there are 768 teaspoons in a gallon.
Reactivity Lab
You are going to set up your multi-well plate as illustrated below.
You need only a few bits of metal for this reaction. One curl of Mg, a 2-3 copper bits and a small pinch of the powdered metals.
For the liquid you only need enough to cover the metal.
Write this down: The color and a brief description of each of the substances you are working with.
Wait 5 minutes
Now describe them again. Is there any change.
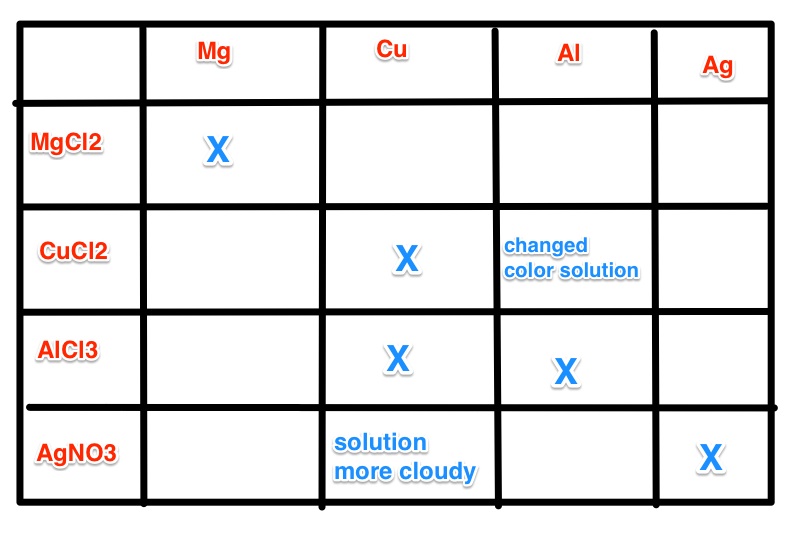
Make a table that describes the changes. Example below.
You are going to set up your multi-well plate as illustrated below.
You need only a few bits of metal for this reaction. One curl of Mg, a 2-3 copper bits and a small pinch of the powdered metals.
For the liquid you only need enough to cover the metal.
Write this down: The color and a brief description of each of the substances you are working with.
Wait 5 minutes
Now describe them again. Is there any change.
Make a table that describes the changes. Example below.
A result section including the table above with your findings and any chemical reactions that occurred need to be turned in. You also need to include a few sentences that are why you believe that some of the wells had reactions but not others.
| iron_sulfide.pdf | |
| File Size: | 37 kb |
| File Type: | |
| atoms_to_moles.docx | |
| File Size: | 44 kb |
| File Type: | docx |
| mole_to_weight.docx | |
| File Size: | 47 kb |
| File Type: | docx |
The problem set below will be due on Tuesday April 22nd.
| percent_composition_and_empirical_formulas.docx | |
| File Size: | 48 kb |
| File Type: | docx |
Hydrates lab The print out
This will be a full lab write up. The result questions should be incorporated into your results table. Remember that your conclusion needs to include why the lab works as well as your error analysis.